What Happened in the Last 24 Hours
On April 22, 2026, SAB Biotherapeutics presented new clinical and mechanistic data from the Phase 1 trial of SAB-142 in adults with established autoimmune Type 1 Diabetes at the International Diabetes Summit (IDS) 2026 (SAB Biotherapeutics via Stock Titan).
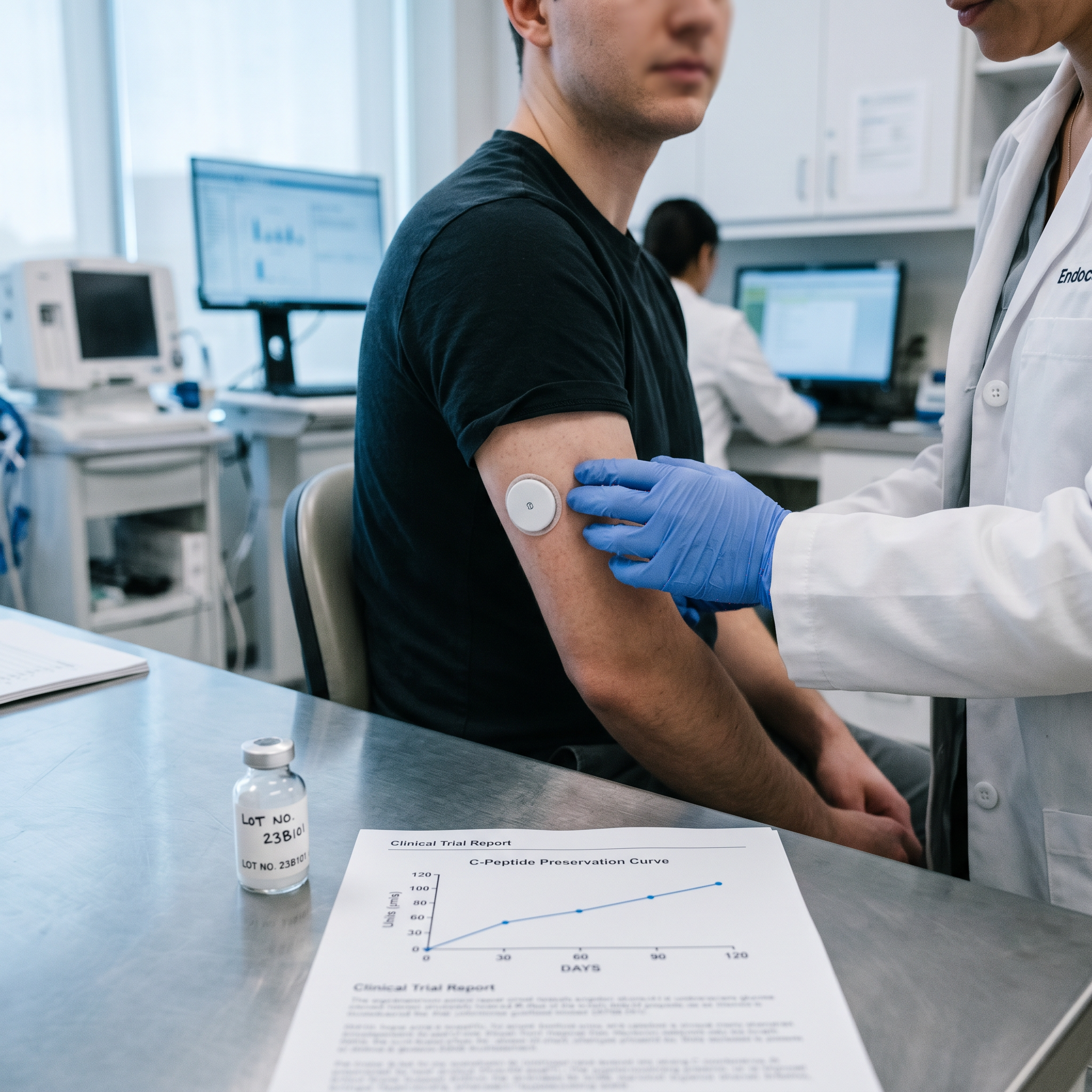
The headline finding: all four SAB-142-treated participants showed preservation of endogenous C-peptide — a biomarker of residual pancreatic beta-cell function — at Day 120, with three of four meeting a pre-specified “super responder” profile. That is a rare signal in established T1D, where C-peptide typically continues to decline after diagnosis.
The Big Takeaways (Fast)
- C-peptide was preserved in 4/4 treated patients. This is an early, small-sample result, but C-peptide preservation is the endpoint regulators and endocrinologists care about in disease-modifying T1D therapy.
- Time-in-range jumped from 73% to 85%. Mean continuous glucose monitor (CGM) time-in-range improved 12 percentage points at Day 120 in the treated cohort.
- Mechanistic support. Investigators observed changes in CD4+ Tconv cell exhaustion (TIGIT+ Tconv levels), consistent with the proposed anti-T-cell mechanism.
- Phase 2b is now the readout to watch. The SAFEGUARD trial (NCT07187531) is the larger follow-up; topline is expected in the second half of 2027.
- This is not a peptide-compounding story. SAB-142 is an IND-stage biologic. It sits in the conventional drug-development pathway, not the 503A/503B compounding debate.
Why This Matters for Peptide Research and T1D
Type 1 Diabetes is the textbook autoimmune disease where the immune system destroys insulin-producing beta cells. Once that process is mostly complete, patients depend on exogenous insulin for life. For decades, the holy grail has been a therapy that slows or halts the autoimmune attack early enough to preserve endogenous insulin production — measured clinically by C-peptide, a fragment released in equimolar amounts with insulin when the body makes its own.
C-peptide is not just a biomarker. Higher residual C-peptide is associated with better glucose control, lower hypoglycemia risk, and reduced long-term complications. That is why the FDA treats C-peptide preservation as a clinically meaningful endpoint — and why a signal in a Phase 1 cohort, even a small one, gets serious attention.
Several peptide-adjacent and antibody-based approaches have tried to move the needle here. Teplizumab (Tzield) is FDA-approved to delay Stage 3 T1D in at-risk individuals. What SAB-142 is attempting is adjacent but distinct: intervening in already-established T1D, where preserving what is left of beta-cell function still translates into real quality-of-life gains.
The Phase 1 Data, in Plain English
According to the IDS 2026 presentation, the Phase 1 cohort included six adults with established Type 1 Diabetes: four received a single dose of SAB-142 at 2.5 mg/kg, and two received placebo (SAB Biotherapeutics).
C-Peptide Preservation at Day 120
All four treated participants showed preserved C-peptide at Day 120 versus baseline — in a disease where C-peptide ordinarily declines progressively. Three of the four met a pre-specified profile investigators labeled “super responder,” meaning their preservation went beyond what would be expected from natural course or background standard of care.
CGM Time-in-Range
Mean continuous glucose monitor (CGM) time-in-range in the treated arm moved from roughly 73% at baseline to 85% at Day 120. For T1D, time-in-range is the single most patient-meaningful glycemic metric — every additional percentage point is associated with lower risk of long-term complications. A 12-point jump in a 120-day window is clinically large, though the cohort is small.
Mechanistic Signal: T-Cell Exhaustion Markers
The investigators also reported changes in TIGIT+ CD4+ Tconv cell populations — an exhaustion marker on the conventional T-cell compartment that is thought to mediate beta-cell destruction in T1D. A rise in exhaustion markers on autoreactive T cells would be consistent with SAB-142’s proposed mechanism of dampening the autoimmune attack. This is the kind of mechanistic anchor that turns a small efficacy signal into a more credible one.
How SAB-142 Compares to Other T1D Biologics
SAB-142 is a polyclonal anti-thymocyte antibody produced in SAB Biotherapeutics’ proprietary transchromosomic cattle platform, designed to target human T cells with reduced immunogenicity compared with rabbit-derived anti-thymocyte globulin (ATG). The program positions it as a next-generation version of the older ATG concept, engineered to be repeatable (patients can be redosed) and more tolerable.
That puts SAB-142 outside the “research peptide” conversation that dominates a lot of PeptideKnow coverage. It is a biologic, developed under an investigational new drug (IND) application, reviewed through the standard CBER/CDER drug-development pathway. It is not compounded, not sold as a research-use-only material, and not implicated in the Section 503A compounding debate we covered yesterday (Peptide Rally Supply Chain and Enforcement Risk).
Why include it on PeptideKnow? Because C-peptide and peptide hormones sit at the center of how the pancreas signals. Therapies that protect residual beta-cell function directly change the calculus for other peptide-adjacent interventions — for example, semaglutide and future GLP-1 / metabolic peptides that work best when a patient still has some endogenous insulin response.
Context: Where the Field Stood Before This Readout
Before this update, the T1D disease-modification field had three kinds of evidence: approved therapies that delay onset in at-risk populations, experimental immunotherapies that produce mixed C-peptide signals in new-onset patients, and essentially nothing credible for established T1D. That last category is where the unmet need is largest — the majority of living T1D patients are years or decades post-diagnosis and have been told, accurately, that no currently available therapy will protect what beta-cell function they have left.
The Phase 1 SAB-142 readout is interesting precisely because it is targeting that hardest-to-treat population. Adults with established T1D were enrolled, not recent-onset cases where residual beta-cell function is inherently higher and outcomes can look good for reasons unrelated to the drug. A positive C-peptide signal in established disease is, mechanistically, a harder result to fake.
Note also the 2.5 mg/kg single dose design. A one-shot intervention producing a detectable biological effect four months later implies either durable receptor-level engagement or a genuine reset of the autoimmune compartment — the TIGIT+ Tconv exhaustion data is consistent with the latter. If that reset is real, the therapy becomes far more commercially and clinically tractable: you do not need chronic weekly dosing forever.
Important Limitations and Caveats
- Tiny sample. Four treated patients, two on placebo. That is appropriate for Phase 1, but absolutely not powered to confirm efficacy.
- No durability beyond Day 120 yet. T1D trials have seen early C-peptide signals that faded by Day 365. Watch for longer-term follow-up before drawing conclusions.
- Single-arm style reporting. Placebo comparator is present but too small to rule out regression-to-the-mean in responders.
- Safety data is early. Anti-thymocyte antibodies have historical safety baggage (cytokine release, infection risk). SAB-142’s engineering is meant to address this, but safety needs a larger denominator.
- Not a cure. Even a best-case outcome here preserves residual function; it does not regenerate beta cells destroyed before treatment. Regenerative approaches are a separate research track.
What to Watch Next
- SAFEGUARD (NCT07187531) topline in 2H 2027. This is the Phase 2b readout that will determine whether the Phase 1 signal survives a properly powered, multi-arm study.
- Longer follow-up on the Phase 1 cohort. Day 180, Day 365, and beyond. If C-peptide preservation holds, the Phase 2b probability of success goes up materially.
- Mechanistic publications. The TIGIT+ Tconv exhaustion finding deserves a peer-reviewed paper. That is how early clinical signals become credible to the broader endocrinology community.
- Regulatory posture. The FDA has shown willingness to engage on T1D disease-modifying therapies (see teplizumab). Breakthrough Therapy designation or a similar expedited path would be a meaningful signal.
PeptideKnow’s Take
This is one of the more clinically consequential peptide-hormone stories of the month, and it has essentially nothing to do with the FDA peptide-compounding news cycle. The SAB-142 Phase 1 data is small, early, and promising — in roughly that order.
If you care about research peptides because you care about metabolic and endocrine biology, this is the kind of data that matters more than another regulatory calendar update. A disease-modifying therapy that preserves endogenous C-peptide in established T1D would reshape how every downstream metabolic intervention is sequenced — including the GLP-1 class and related agents.
Our read: cautious optimism, and mark 2H 2027 on the calendar.
Frequently Asked Questions
What is SAB-142?
SAB-142 is a polyclonal anti-thymocyte antibody developed by SAB Biotherapeutics, produced in transchromosomic cattle to be fully human. It is being developed as a disease-modifying therapy for Type 1 Diabetes, aiming to dampen the autoimmune attack on pancreatic beta cells.
What is C-peptide and why does it matter?
C-peptide is a fragment released alongside insulin when the pancreas makes its own insulin. In Type 1 Diabetes, C-peptide levels fall as beta cells are destroyed. Preserving residual C-peptide is associated with better glucose control, fewer hypoglycemic events, and reduced long-term complications — which is why the FDA recognizes it as a clinically meaningful endpoint.
How big was the Phase 1 trial?
The Phase 1 adult T1D cohort included six participants: four received SAB-142 at 2.5 mg/kg and two received placebo. Small numbers are typical for Phase 1 safety and proof-of-mechanism work.
Is SAB-142 a peptide?
No. SAB-142 is a biologic — specifically a polyclonal anti-thymocyte antibody. It is developed through the standard FDA drug pathway and is not related to the Section 503A peptide compounding debate around peptides such as BPC-157 or TB-500.
When will we know if this works?
The Phase 2b SAFEGUARD trial (NCT07187531) has a topline readout expected in the second half of 2027. Longer-term follow-up from the Phase 1 cohort (Day 180 and beyond) may arrive sooner and will indicate whether the initial C-peptide signal is durable.
Does this mean T1D will have a cure soon?
No. Even a successful outcome would preserve residual beta-cell function — it would not regenerate beta cells already destroyed. A true cure likely requires a combination of immune modulation (like SAB-142) and beta-cell replacement or regeneration.
Sources
- SAB Biotherapeutics Presents Additional Clinical and Mechanistic Data from SAB-142 Phase 1 Trial at IDS 2026 — April 22, 2026
- ClinicalTrials.gov: SAFEGUARD Phase 2b Study (NCT07187531)
- FDA: Teplizumab (Tzield) approval to delay onset of Type 1 Diabetes
- JDRF: Why Endogenous Insulin Production (C-Peptide) Matters in T1D
- Diabetes Care: Clinical Targets for Continuous Glucose Monitoring (Time-in-Range Consensus)
Sources & References
- FDA PCAC Meeting Announcement (July 23-24, 2026)
- PBS: FDA to Weigh Easing Limits on Peptides Favored by RFK Jr.
- BioPharma Dive: FDA Peptides RFK Advisory Committee Restrictions
- RAPS: FDA Considers Adding a Dozen Peptides to Bulk Drug List
- Ars Technica: RFK Jr. Forces FDA to Reconsider 12 Peptides
- ProPublica: Peptide Safety Investigation
- New York Times: Peptide Ban FDA RFK Jr.
- SSRP Institute: FDA Announces Change in Status of 12 Peptides
- CNBC: RFK Jr. Peptides Hims Hers GLP-1
- USA Today: RFK Jr. FDA Peptides Explainer